Clinical Trials
LACOG 0415
Title
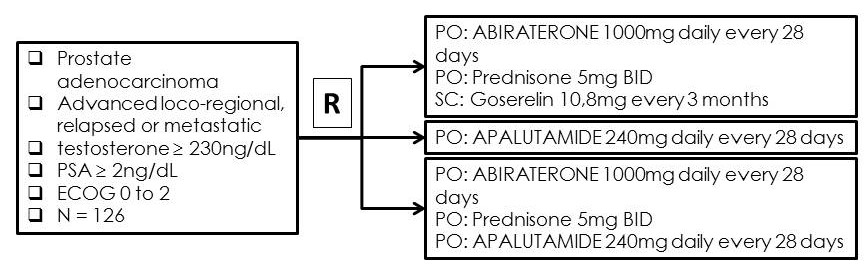
Phase II randomized study of Abiraterone acetate plus ADT versus APALUTAMIDE versus Abiraterone and APALUTAMIDE in patients with advanced prostate cancer with non-castrate testosterone levels
Primary Objective
We aim to assess the activity, safety and patients reported outcome of ADT plus abiraterone, abiraterone plus APALUTAMIDE (a second-generation antiandrogen) or APALUTAMIDE alone in hormone naïve locally advanced or metastatic prostate cancer which ADT was indicated.
Design
Sample size: 126
Study Scientific Committee: Fernando Maluf, Oren Smaletz, Fábio Schutz, Vinicius Carreira, André Fay,Daniel Herchenhorn,Telma Santos.
Status: Regulatory process
Sponsor: Janssen
Registry
NCT02867020
Contact
lacog@lacog.org.br
55 51 33845334
